INTRODUCTION
Glaucoma is a chronic, progressive optic neuropathy and a leading cause of irreversible blindness worldwide. Sustained reduction of intraocular pressure (IOP) remains the only proven strategy to slow the progression of the disease. While pharmacological and laser therapies are considered first-line treatments, many patients ultimately require surgical intervention to achieve target IOP levels.
Trabeculectomy remains the gold standard in glaucoma surgery due to its ability to produce substantial and long-lasting IOP reduction [1]. However, it is associated with significant risks, including hypotony, bleb leaks, and choroidal detachment, as well as the need for intensive postoperative care [2]. These limitations have led to increasing interest in less invasive surgical alternatives [3].
Minimally invasive micro-sclerostomy (MIMS) is a novel ab interno filtration procedure that creates a transscleral fistula to facilitate aqueous outflow into the subconjunctival space. In contrast to other minimally invasive glaucoma surgeries (MIGS), which typically enhance conventional or uveoscleral outflow, MIMS enables subconjunctival filtration without the need for permanent implants. This approach may reduce long-term complications such as implant migration, fibrosis, or chronic tissue responses [4, 5]. Initial clinical studies suggest that MIMS provides meaningful IOP reduction with a favorable safety profile [6–8]. However, further research is needed to define its role within the surgical treatment algorithm for OAG. The present study aimed to evaluate the one-year efficacy and safety of MIMS in patients with OAG who remain uncontrolled despite maximal tolerated medical therapy.
MATERIAL AND METHODS
This prospective interventional case series included 17 eyes of 17 patients diagnosed with OAG, in whom IOP remained uncontrolled despite maximally tolerated topical therapy. Patients were eligible for inclusion if they were 18 years of age or older, had an open iridocorneal angle confirmed by gonios-copy, and had documented progression or persistently elevated IOP despite the use of at least two topical antiglaucoma medications. Exclusion criteria included angle-closure glaucoma, previous incisional glaucoma surgery in the study eye, active intraocular inflammation or infection, and significant coexisting ocular disease, such as advanced macular degeneration. All patients underwent ab interno micro-sclerostomy using the MIMS technique, performed by experienced glaucoma surgeons. The procedure was carried out under topical anesthesia. A 1.5 mm temporal paracentesis was created, and approxi- mately 0.2 ml of ophthalmic viscoelastic device (Viscoat) was injected into the anterior chamber to maintain space. Subsequently, an ophthalmic viscoelastic agent was injected into the superonasal subconjunctival space to facilitate tissue elevation and accommodate the advancement of the surgical device. A subconjunctival injection of 0.1 ml of mitomycin C (0.3 mg/ml) was then administered to modulate wound healing and reduce fibrosis formation [6–8].
The proprietary MIMS device was introduced into the anterior chamber and positioned at the superior nasal angle. Using a controlled rotational mechanism, a transscleral micro- sclerostomy tunnel was created, allowing aqueous outflow into the subconjunctival space [6, 8]. The excised scleral tissue cylinder (approx. 0.3 mm) was removed via the lumen of the device. No stent or implant was left in place.
After confirming bleb formation and percolation, the anterior chamber was partially irrigated to remove excess viscoelastic. Ciprofloxacin 0.3% was instilled at the end of the procedure. No sutures were used.
Postoperatively, all patients received a standard regimen of topical corticosteroids and antibiotics, tapered gradually over a 4- to 6-week period. Follow-up examinations were performed at five time points: on the first postoperative day, and at 1, 3, 6, and 12 months after surgery. At each visit, best-corrected visual acuity was assessed, IOP was measured with Goldmann applanation tonometry, and the number of prescribed antiglaucoma medications was recorded. Any postoperative complications – such as bleb failure, hypotony, or transient iris occlusion of the sclerostomy site – were noted. The primary endpoints of the study were the mean change in IOP and the mean change in the number of antiglaucoma medications from baseline to 12 months postoperatively. Secondary outcomes included the rate of complete surgical success, defined as IOP ≤ 18 mmHg with a reduction of at least 20% from baseline and no use of medications, and qualified surgical success, defined as IOP ≤ 18 mmHg with a ≥ 20% reduction from baseline regardless of medication use. Additional outcomes included the proportion of patients who achieved medication independence and the incidence of intraoperative or postoperative complications. Continuous variables were presented as mean values with standard deviations (mean ±SD). Pre- and postoperative values were compared using paired t-tests. A p-value of < 0.05 was considered statistically significant. The study protocol was approved by the institutional ethics committee and adhered to the tenets of the Declaration of Helsinki. Written informed consent was obtained from all participants.
RESULTS
Seventeen eyes of 17 patients with OAG were included in this prospective study. At baseline, the mean IOP was 20.5 ±3.5 mmHg, and patients were using an average of 2.7 ±0.9 antiglaucoma medications.
By 12 months postoperatively, the mean IOP had decreased significantly to 16.0 ±6.6 mmHg, reflecting a 22% reduction from baseline (p = 0.0019, paired t-test). Concurrently, the mean number of medications declined to 0.71 ±0.99, corresponding to a 74% decrease (p < 0.001).
Complete surgical success – defined as IOP ≤ 18 mmHg with at least a 20% reduction from baseline and no need for antiglaucoma medications – was achieved in 41.2% of eyes (7/17). Qualified surgical success – meeting the same IOP criteria regardless of medication use – was observed in 58.8% of cases (10/17). In addition, 58.8% of patients (10/17) were medication-free at 12 months.
No intraoperative complications were reported. Postoperative transient iris occlusion at the sclerostomy site occurred in 3 eyes (17.6%) and was successfully treated with pilocar-pine and laser iridoplasty. No instances of hypotony, choroidal detachment, bleb failure, or endophthalmitis were observed throughout the follow-up period.
These findings support the efficacy and safety of MIMS as a minimally invasive, implant-free surgical technique for lowering IOP and reducing reliance on medications in patients with OAG.
DISCUSSION
Trabeculectomy, historically attributed to Cairns [1], remains the most effective surgical method for long-term IOP reduction but is associated with significant complications such as hypotony, bleb-related issues, and intensive postoperative management [2].
This study demonstrates that MIMS is a safe and effective surgical option for reducing IOP in patients with OAG inadequately controlled despite maximal medical therapy. At 12 months postoperatively, we observed a significant reduction in both IOP and the number of antiglaucoma medications, without any intraoperative complications or serious postoperative events.
Our findings are in line with those of Voskanyan et al. [6], who reported outcomes from a multicenter prospective study of 120 eyes with primary or pseudoexfoliative OAG undergoing MIMS. They observed a 38% IOP reduction (from 27.9 ±3.7 mmHg to 17.5 ±5.3 mmHg) and surgical success rates of 70.5% (complete) and 82.1% (qualified). In our cohort, IOP was reduced by 22% (from 20.5 to 16.0 mmHg), with Minimally invasive micro-sclerostomy (MIMS): a novel approach to lowering intraocular pressure and reducing medication burden
complete and qualified success achieved in 41.2% and 58.8% of eyes, respectively. The more modest outcomes in our study may be attributed to the lower baseline IOP and the use of more stringent criteria for surgical success (IOP ≤ 18 mmHg versus ≤ 21 mmHg in the Voskanyan study).
Compared to traditional filtration procedures such as trabeculectomy [1, 2], MIMS offers a significantly less invasive surgical approach (Table I) while still enabling aqueous outflow into the subconjunctival space. Trabeculectomy, though highly effective, requires conjunctival and scleral dissection, suture management, and intensive follow-up, and carries risks such as bleb leaks, hypotony, and choroidal ef-fusion. MIMS, in contrast, is performed ab interno through a corneal incision, requires no implant or conjunctival manipulation, and preserves the superior conjunctiva for future interventions. Although the IOP-lowering effect of MIMS may be somewhat less pronounced, its lower complication rate and surgical simplicity make it an attractive alternative in selected cases.
Table I
Comparative characteristics of bleb-forming glaucoma surgical techniques: minimally invasive micro-sclerostomy (MIMS), XEN Gel Stent, Preserflo MicroShunt, and trabeculectomy
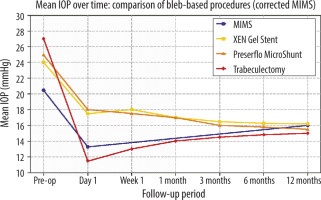
MIMS also compares favorably to other subconjunctival MIGS procedures, such as the XEN Gel Stent and Preserflo MicroShunt [4, 5]. The comparative IOP outcomes are presented in Figure 1. Like these devices, MIMS facilitates filtration through the subconjunctival route, but avoids implanting a permanent foreign body. The absence of an implant eliminates risks such as migration, exposure, and foreign-body reaction. Additionally, MIMS avoids issues related to implant positioning and may reduce the need for postoperative interventions such as needling or revisions. While the XEN Gel Stent is delivered ab interno, precise placement can be technically demanding. Preserflo, inserted ab externo, requires conjunctival dissection and carries the typical risks of implant-based bleb surgery. In contrast, canal-based MIGS procedures such as the Kahook Dual Blade and GATT [9, 10] do not create a filtering bleb and aim to enhance conventional trabecular outflow through Schlemm’s canal. Similarly, implant-based devices like the iStent or Hydrus microstents [11, 12] facilitate trabecular outflow but remain confined to the anterior chamber and do not bypass the conventional outflow system via subconjunctival filtration. MIMS thus provides a simplified, stent-free approach with a similar outflow pathway to XEN or Preserflo, yet with potentially fewer implant-related complications.
Figure 1
Mean intraocular pressure (IOP) at baseline and 12 months following various bleb-forming glaucoma procedures: MIMS (minimally invasive micro-sclerostomy; present study), XEN Gel Stent [2, 3], Preserflo MicroShunt [4], and trabeculectomy [5, 6]. Data for MIMS are derived from the current study; comparative values were obtained from peer-reviewed publications
With respect to safety, the most common postoperative event in our study was transient iris occlusion of the sclerostomy site, observed in 17.6% of cases. All instances were successfully managed with pilocarpine and laser iridoplasty. Importantly, no cases of hypotony, choroidal detachment, bleb failure, or endophthalmitis occurred. These findings are consistent with previously reported safety profiles for MIMS [6–8].
Overall, MIMS represents a unique category of bleb- forming glaucoma surgery – offering the benefits of ab interno access, implant-free design, and preservation of conjunctival integrity. This combination of safety, efficacy, and technical simplicity makes it a compelling option for patients requiring moderate IOP lowering, particularly those at risk of implant-related complications or in whom conventional filtration is less desirable.
CONCLUSIONS
MIMS demonstrated a favorable safety and efficacy profile in this prospective study of patients with OAG whose IOP remained uncontrolled despite maximal topical therapy.
The procedure resulted in significant and sustained IOP reduction, along with a marked decrease in the need for anti-glaucoma medications. No serious intraoperative or postoperative complications were observed, and more than half of the patients achieved medication independence at 12 months.
Within the spectrum of bleb-forming procedures, MIMS occupies a distinct position. Unlike subconjunctival MIGS techniques such as the Preserflo MicroShunt or XEN Gel Stent [4, 5], MIMS enables aqueous humor outflow via a controlled transscleral fistula without introducing an implant. The absence of a foreign body reduces the risks of migration, erosion, chronic inflammation, or fibrotic encapsulation. Moreover, by preserving the conjunctiva and minimizing surgical trauma, MIMS offers the technical advantages of ab interno approaches while maintaining meaningful IOP-lowering efficacy [6, 7].
Given its balance of safety, simplicity, and effectiveness, MIMS may serve as a valuable bridging option between conventional filtering surgery and implant-based MIGS. Further randomized studies with larger cohorts and extended follow-up are warranted to validate these findings and to better define the role of MIMS in the surgical management of glaucoma [6–8].
STUDY LIMITATIONS
This study has several limitations. The relatively small sample size and absence of a control group may limit the generalizability of the findings. In addition, the 12-month follow- up period, although clinically relevant, does not provide insights into long-term outcomes, including the durability of IOP control or late complications. Nevertheless, the observed IOP and medication reductions align with prior reports and support the clinical utility of MIMS [6, 7]. Future randomized, controlled studies with larger cohorts and extended follow-up are warranted to confirm these results and better establish the role of MIMS in glaucoma surgery [6–8].







