INTRODUCTION
Around 200 million people worldwide suffer from age- related macular degeneration (AMD) [1]. New cases are on the rise due to aging of the population. Studies show that the risk of early AMD is 25% and the risk of late AMD is 8% in patients over the age of 75 [2]. As many as 10% of patients with dry AMD eventually develop choroidal neovascularization and the wet form of the disease develops [3]. Wet (exudative, neovascular) AMD is one of the most common causes of severe visual impairment or even total blindness in the population over 60 years of age [1, 3].
Anti-vascular endothelial growth factor (anti-VEGF) therapy remains the only mode of treatment of wet AMD; however, for various reasons this treatment may be delayed or omitted, leading to fatal complications such as submacular haemorrhages.
Submacular haemorrhage (SMH) is a rare but potentially damaging phenomenon. The natural course of this complication leads to complete blindness and usually to formation of a fibrous scar in the macular region [4]. Subretinal blood causes damage of photoreceptors as soon as 24 hours after the onset of haemorrhage. It also acts as a mechanical barrier for photoreceptors and induces toxic damage (hemosiderin) to them. Contraction forces which arise during scar formation may cause problems in the macular region [5].
It seems that removing the haemorrhage from the macular region is the key step to restore the function of photoreceptors; however, there is no accepted gold standard treatment of submacular haemorrhage.
The aim of the study was to evaluate the outcome of pars plana vitrectomy, subretinal injection of tissue plasminogen activator (tPA), anti-VEGF bevacizumab injection into the vitreous cavity and partial fluid-air exchange in patients with submacular haemorrhage (SMH) secondary to wet AMD.
MATERIAL AND METHODS
Charts of 10 patients operated on between January 2021 and June 2022 were reviewed. The inclusion criterion for the surgical intervention was thick haemorrhage (no underlying structure seen) located in the macular region which was at least 1.5 discs in diameter.
The surgical procedure included 23-ga vitrectomy, administration of subretinal tPA in the concentration of 12.5 µg/0.1 ml (total given volume up to 0.2 ml), anti-VEGF bevacizumab 1.25 µg injection into the vitreous cavity and partial fluid-air exchange (partial in this case means that part of the vitreous cavity was filled with air). The tPA administrations into the subretinal space were done with a 41-ga cannula (by Dorc). No laser treatment of retinal paracentesis was necessary. Patients were kept in a horizontal position for 30 minutes after the surgery to allow tPA to reach the subretinal space. No other positioning was required after the procedure.
The procedure was performed as soon as possible after the vitreo-retinal consultation.
The delay in seeking the ophthalmic consultation was different for different patients. Some of them were waiting for the problem to resolve spontaneously, some had problems with general ophthalmic consultation, and some were told by the general ophthalmologist that no treatment was possible in the case of submacular haemorrhage.
RESULTS
The observation period for all patients was 6 months. Ten patients were enrolled in the study: 6 female and 4 male. The age of the patients was from 71 to 81 years, with the mean of 75.6 years. All the patients had SMH caused by wet AMD. The patients’ characteristics are shown in Table I.
Table I
Patients’ characteristics
In 4 patients the SMH was the first sign of wet AMD. In 6 patients the eye which experienced the haemorrhage had been treated with anti-VEGF previously. The number of injections varied from 8 to 20. The anti-VEGF medications which were used before the SMH occurrence were aflibercept in 4 cases and bevacizumab in 2 cases. There was no patient with SMH who was treated with ranibizumab.
In 5 patients the eye with SMH was the only functioning eye of those patients. The fellow eye was lost due to advanced wet AMD (patients 1, 3, 5 and 9). Patient 3 had been treated previously in another clinic for pneumatic SMH displacement. Bevacizumab and 20% SF6 were given intravitreally. No improvement with this treatment was noted.
In 2 cases haemorrhage occurred in both eyes almost simultaneously and it was decided to operate on the eye with more severe haemorrhage (patients 4 and 6). Only 2 patients in the study were on anticoagulant therapy and both had been taking aspirin orally before the haemorrhage occurred.
Anticoagulant therapy was discontinued in those cases soon after the onset of haemorrhage (patient 3 and 7).
Surgery was performed between day 7 and day 42 after the occurrence of haemorrhage. The mean time was 19.6 days. The difference in the timing of surgery was due to late presentation of some patients at the clinic. Despite late presentation, patients 2, 3, 4 and 9 underwent the surgery in the usual manner. In patients 3 and 9 the operated eye was the only functioning eye so the surgery was the only chance for them to regain some visual acuity.
Visual acuity at the time of the surgery was low in all patients and ranged between 0.05 and hand movement (HM). In all patients visual acuity (VA) improvement was seen after surgery. VA ranged from 0.05 to 0.8. The mean VA after surgery was 0.35. The best VA improvement was seen in patient 1, who was operated on 7 days after the onset of the haemorrhage, and who was previously treated with anti-VEGF (aflibercept). The worst VA was achieved in patient 2, who had not been treated before and who was operated late – 35 days after the haemorrhage onset.
Patient 1
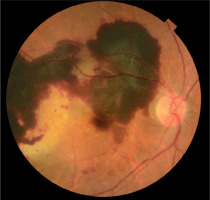
A 75-year-old woman was seen in the clinic due to a sudden VA deterioration in her right eye. The examination revealed an SMH (Figure 1). The eye had previously been treated for wet AMD and had received 9 injection of aflibercept. VA in the right eye was 0.05.
Her left eye had low visual acuity of hand motion. It was diagnosed with wet AMD. On examination a scar in macular region was seen.
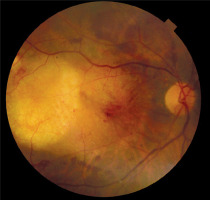
The patient suffered from hypertension and was a heavy smoker. She was not on anticoagulant treatment. The surgery was performed on day 7 after the SMH occurrence. No complications were noted during or after the surgery. Figure 2 shows a fundus photograph after the surgery. After surgery the patient was put on a month of bevacizumab injections into her right eye.
Three months after surgery VA in the right eye was 0.8.
Patient 3
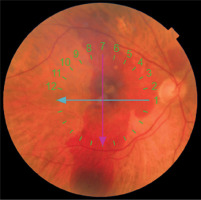
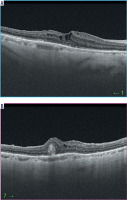
A 71-year-old woman was seen due to an SMH in the right eye. VA in the right eye was HM. Figure 3 shows the fundus of the right eye. Figure 4 shows OCT of the right eye.
Figure 4
Optical coherence tomography (OCT) examinations of RE of patient 3 before vitrectomy. Different scans of the same eye. Elevation shows the blood under the retina
She suffered from diabetes mellitus type II, hypertension and was on aspirin once daily. According to the patient, the SMH had occurred several weeks earlier and the patient had been treated with intravitreal bevacizumab and 20% SF6 to displace the haemorrhage from macular region. The technique was unsuccessful.
The left eye of this patient was lost due to wet AMD. Both eyes had signs of minor background diabetic retinopathy which needed no treatment.
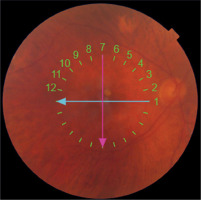
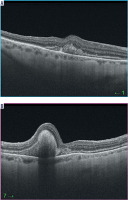
Surgery was performed on day 42 after SMH onset and was uneventful. VA improvement was achieved and 3 months after surgery was 0.3. Figure 5 shows the fundus and Figure 6 shows OCT of the RE a month after surgery.
DISCUSSION
Submacular haemorrhage (SMH) is a vision-threatening condition. The natural history of SMH described clearly gives no chance to keep useful visual acuity [4, 5]. Blood under the central retina has a toxic effect on photoreceptors and left in such a location causes a decline in visual acuity to the level of hand motion. Therefore prompt surgical treatment remains the best option. Surgery should be performed as soon as possible; however, no study has been carried out to define the time limits which guarantee the best results.
Various treatment methods have been proposed including anti-VEGF treatment. There are methods involving gas displacement of a previously dissolved clot which uses SF6 gas and tPA applied to the vitreous cavity. There are publications on use of subretinal injection of tPA during vitrectomy. There is still no gold standard treatment for SMH [6].
Use of tPA to mobilise the submacular blood which clots soon after spreading away from vessels seems to be essential to achieve the necessary outcome [5, 7-9]. In 1996, Kamel et al. used tPA to remove a subretinal haemorrhage for the first time; however, the main mechanical action was performed using perfluorocarbon [7]. Pneumatic displacement of the SMH was first described by Wilson J. Heriot in 1996. Heriot’s initial technique involved intravitreal tPA injection and subsequent haemorrhage displacement by gas injected into the vitreous cavity [8]. Since this initial report, several small series have been described.
Pneumatic haemorrhage displacement can be performed as an office procedure under local anaesthesia (it is done in the USA where no operating room is required to perform ocular injections – author’s observation). There is no need to prepare the patient for extensive surgery, so the intervention can be performed almost immediately after the diagnosis is made. The dosage of tPA used for pneumatic haemorrhage displacement varies between 25 and 50 µg/ml [7-9]. The toxic dose of tPA is considered to be 50 µg/ml and should not be exceeded during any procedure [7-9]. The amount of tPA applied into the vitreous cavity during the pneumatic clot displacement should be, however, large enough to achieve the therapeutic drug level under the retina. There are no studies evaluating in vivo tPA permeability through the human retina, so we do not know for sure how much drug reaches the subretinal space. Pneumatic displacement is associated with three main complications: acting without control because the drug is not administered directly under the retina, creating retinal breaks, and increasing intraocular pressure, which should be managed by paracentesis [7-9].
Additionally, the patient must adopt a position after the procedure. First, they should remain in a horizontal position to allow tPA to sink to the bottom of the eye and penetrate through the retina to the submacular region and allow liquefaction of blood, and then they must keep their face 45 degrees down to allow gas to move the clot down away from the macular region [4, 7-9].
Subretinal tPA injection seems to be a more controlled and precise procedure. The dose of the medication that is used may be reduced to the concentration of 12.5 µg/0.1 ml as it is administered directly into the subretinal space where the clot is situated. The volume given under the retina never exceeds 0.2 ml, which gives 25 µg in total, which is less than the toxic level. Patients are placed in a horizontal position for 30 minutes after the surgery to ensure that the tPA remains in the submacular space.
All the surgical complications which may occur while administering any substance into the eye, such as retinal breaks, holes, retinal detachment, and high intraocular pressure, are much better controlled during vitrectomy. They may be addressed during the same procedure so no complications would be seen in the postoperative period.
There are several modifications of vitrectomy and submacular tPA administrations. Some authors describe adding filtered air to the tPA solution and applying the mixture subretinally to facilitate haemorrhage liquefaction and displacement. After administration under the retina, air travels up and pushes the liquefied blood down away from the macular region when the patient is in an upright position [4].
When tPA was used as the sole therapeutic agent, subretinal haemorrhage resolved during 24 to 48 hours. The author believes that air injection is not absolutely necessary for blood absorption, thus allowing for reduction of the volume of the mixture given under the retina and, possibly, leading to less damage. Another surgical modification consists in the use of anti-VEGF during the procedure. If the haemorrhage is caused by wet AMD, anti-VEGF is the treatment of the underlying condition.
Authors usually present two main methods of administering anti-VEGF: under the retina or together with tPA and air [7-9].
Usually, bevacizumab is the medication of choice to be administered during the procedure since none of the registered anti-VEGF medications are registered to be given during the surgery.
Intravitreal anti-VEGF seems to be a safer procedure since we are not sure about its toxicity for the retina and RPE. The results of this study are consistent with other published series [4, 7-9]. Better visual acuity was achieved in eyes which were operated on soon after haemorrhage onset since the damage of the photoreceptors was probably smaller.
It should be emphasised that visual acuity outcome after the surgery also depends on the state of the macula before the onset of the haemorrhage.
This study also presents rarely published data concerning patients operated on for SMH late. Late surgery was not the action of choice in those cases but rather was the consequence of a series of unfortunate coincidences. However, performed late, the surgery helped eliminate the haemorrhage from the subretinal space and improved visual acuity. Having in mind all the limitations of the study, a small group and different surgery timing, the described trial should be recognised as a case series of a specific surgical approach. It is very difficult to compare the results in patients covered by the study, because their macula condition was different before the surgery. The timing of the surgery was also different.
As long as no major complications were noted after the surgery in our case studies, having in mind the unfavourable natural history of SMH, surgery with subretinal tPA might be considered as the treatment of choice.
No case series, however, proves that a method will be successful. In the case of submacular haemorrhage, published data usually come from small case series. No blinded study with a large number of patients is available at the moment in this field.
CONCLUSIONS
Submacular haemorrhage (SMH) in wet AMD is a sight-threatening phenomenon which should be addressed as soon as possible. To date there is no established mode of treatment of those patients. Different authors propose different approaches. Surgical intervention – vitrectomy, subretinal tPA injection, anti-VEGF injection into the vitreous cavity and partial fluid-air exchange – is an effective procedure to achieve visual acuity improvement.
Due to the rare occurrence of SMH, no study performed on a large population is available to evaluate the proper timing of the surgical intervention.
This study describes another series of patients operated on with tPA given subretinally. The results of this case study are similar to those of other authors.
What is new in this study is that some patients were operated on late due to SMH (more than a week after SMH occurrence). To the author’s best knowledge, such late intervention has not been reported before.
Even if performed late, surgery in all patients was successful to the degree allowed by the state of the macula.