INTRODUCTION
Topical non-steroidal anti-inflammatory drugs (NSAIDs) are commonly prescribed after different surgical and laser procedures in ophthalmology. The main goal is to decrease the inflammation and pain as well as prevent macular edema [1]. There are 4 classes of NSAIDs and each of them has different action on cyclooxygenase (COX) responsible for inflammatory mediator synthesis. Diclofenac belongs to class 2 because it inhibits COX-1 and COX-2. Action regarding COX-3 is not known. Additionally, diclofenac inhibits lipoxygenase; therefore its action is similar to steroids, but less potent. NSAIDs used in ophthalmology usually cause mild secondary side effects, i.e. local burning, itching, hyperemia and superficial punctate keratitis. Severe complications of corneal melt and corneal perforations have been very rarely reported [2-5].
CASE REPORT
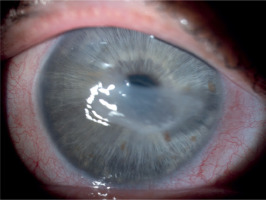
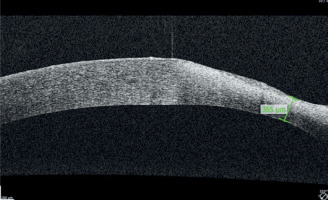
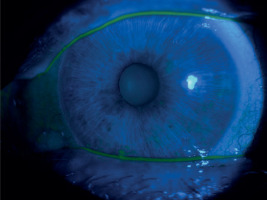
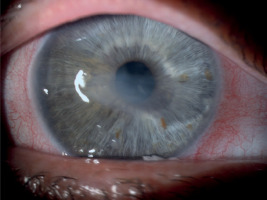
A 65-year-old woman, with a history of diabetes on oral medication for 5 years but no signs of diabetic retinopathy, was operated on for cortico-nuclear cataract by phacoemulsification with IOL PC with no perioperative complications. Her Snellen best corrected visual acuity (BCVA) was 0.6 preoperatively. The eye was quiet on the day of surgery; however, per records slight bilateral conjunctival redness with some secretion was noted 2 weeks before the operation. This condition was managed by prescription of Tobradex (tobramycin + dexamethasone) for 1 week. No BUT/fluorescein staining was performed at that time. Immediate postoperative BCVA was 0.5. The patient was discharged from the hospital with routine prescription of antibiotic-steroid drops 4 times a day and 0.1% diclofenac drops 4 times a day (Dicloabak, Laboratoires THEA, France). One week after the operation she presented to the emergency room complaining of foreign body sensation without any pain. Her BCVA was CF at 1 m. Slit lamp examination showed little perilimbal injection with horizontally oval corneal ulcer located centrally (Figure 1). On OCT decrease in thickness of the cornea corresponding to this ulcer was noted (Figure 2). The other eye showed signs of filamentary keratopathy (Figure 3). The serious nature of the complication was discussed with the patient. Diclofenac and steroid drops were stopped. Nonpreservative tears hourly and levofloxacin drops 3 times a day were prescribed. Successful healing of corneal ulcer was noted at subsequent follow-up visits at 1 week, 2 weeks and 1 month without any need for amniotic membrane transplantation. At the 1-month follow-up visit her BCVA was 0.4 and there was opacification at the anterior cornea and persistent filamentary debris (Figure 4). Further investigations revealed increased ESR and circulating autoantibodies SS-A, SS-B, Ro-52. In addition, the patient’s history was significant for mouth dryness and lack of smell for about 7 years. Subsequently, she has been diagnosed with Sjögren syndrome.
DISCUSSION
This report describes a case of stage 3 neurotrophic keratitis likely related to diclofenac [6]. It occurred after an uneventful cataract operation when the drug was prescribed according to recommendations to prevent macular edema in a diabetic patient. As the patient suffered from filamentary keratopathy associated with primary Sjögren syndrome, the use of diclofenac should have been excluded. In the case of filamentary keratopathy the corneal epithelium suffers from severe dryness and damaging mechanical movements of filaments. Although Dicloabak does not contain conservatives which are toxic for the cornea such as benzalkonium chloride (BAK), its use leads to a decrease of corneal sensitivity. Also the corneal incisions contribute to hypoesthesia. Hypoesthesia dysregulated further tear production in our predisposed patient. As a consequence the hyperosmolarity of the tears activates the inflammatory cells/inflammatory cytokines, which are responsible for the melting process of the corneal stroma [7].
Obviously one can argue that steroids and diabetes also have to be considered in our patient as a cause of corneal ulcer; however, the very short time between the prescription and corneal melting makes its unlikely. In fact the patient noticed the visual loss 5 days after the operation so the corneal ulcer was already there. As we know, occasionally steroids are prescribed for a short duration (1-3 weeks) in cases of dry eye to relieve the inflammatory factor, and it does not usually cause any problem. If that occurs, steroids prevent healing after a longer period. On the other hand, diabetes could be responsible for sensitive neuropathy but usually after many years if not well managed [8].
In a series of 8 cases of severe corneal complications after topical indomethacin use, two described patients did not take any steroids. The first one experienced corneal perforation 11 days after cataract surgery with tropicamide as the only concomitant medication and the second one experienced ulceration 31 days after cataract surgery with tobramycin, rifamycin and non-preserved tears as the concomitant medications [4].
In addition, diclofenac (commercial name of Falcon) has already been reported to cause melting of the cornea. After several adverse events reported to the American Society of Cataract and Refractive Surgery, the drug was withdrawn from the US market [7].
Accordingly, in our report, diabetes and steroid use were considered unlikely to be a major factor in ulcer development but perhaps were only aggravating factors.
We believe that the prescription of NSAIDs should be preceded by detailed questioning of the patient about predisposing conditions such as rheumatoid conditions and careful examination of the eye to exclude severe dry eye syndrome. Following the surgery patients with worsening vision should come for evaluation to the emergency department without delay.